RESEARCH HIGHLIGHT: Targeted gene insertion in the AAVS1 safe harbor site helps identify why hPSCs are recalcitrant to CRISPR/Cas9 genome editing
Without a doubt, CRISPR/Cas9 technology is one of the most exciting and promising molecular biology tools of our time.
But some cell lines, like human pluripotent stem cells (hPSCs), have been difficult to engineer. In an insightful study published in the June 11, 2018, issue of Nature Medicine, Ihry, et al.1 show why hPSCs are resistant to genome engineering by CRISPR/Cas9 and, in the process, they raise a few caveats for the use of CRISPR/Cas9 technology to engineer hPSC-based therapies.
p53 inhibits CRISPR–Cas9 engineering in human pluripotent stem cells
Ihry RJ, et al. Nature Medicine. 11 June 2018. 24:939–946. DOI: 10.1038/s41591-018-0050-6
The Challenge: hPSCs have been resistant to genome engineering using CRISPR/Cas9.
The Advances: Ihry, et al,1 were able to create a line of hPSCs that showed an average rate of over 80% Cas9-mediated insertion/deletion. They achieved this high engineering success rate through the use of an inducible Cas9 gene inserted at the adeno-associated virus integration site 1 (AAVS1), also known as the AAVS1 Safe Harbor Site. Unfortunately, Ihry, et al, also found that while they were able to consistently generate their desired genome edits, the majority of hPSCs died. They found that DSBs generated by Cas9 are particularly toxic to hPSCs, and that this toxicity is dependent on p53.
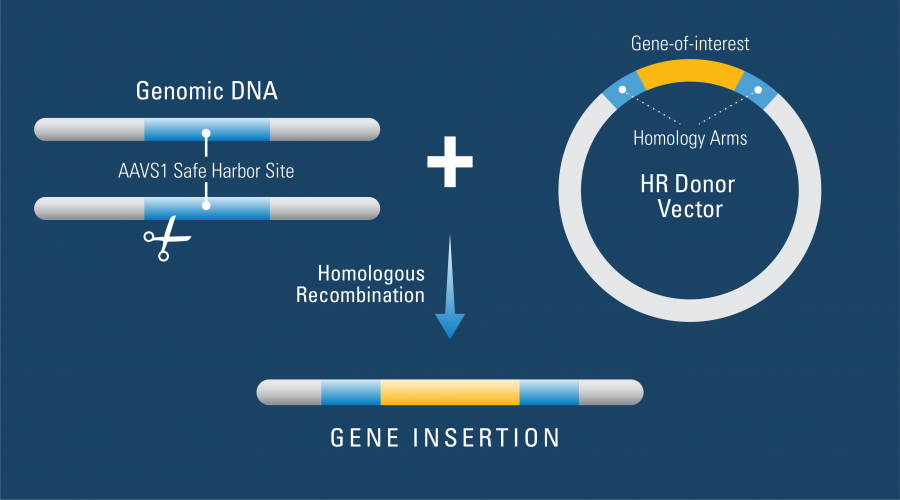
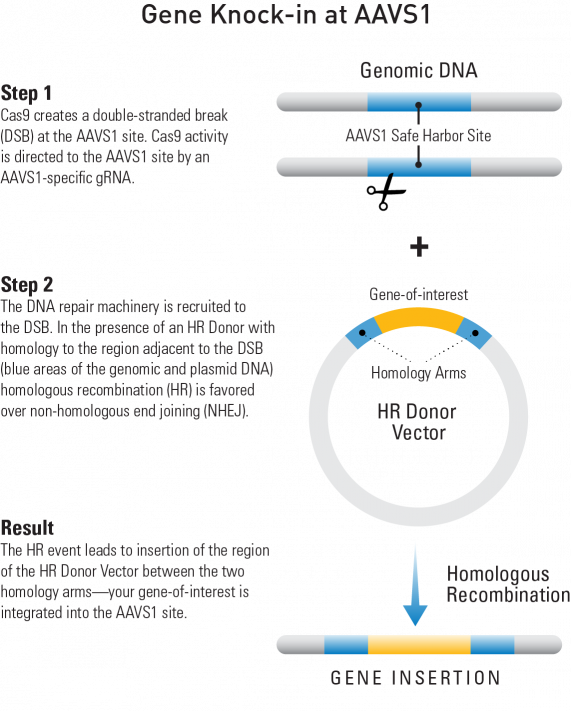
How SBI Helped: To create their line of hPSCs with an inducible Cas9 gene stably integrated into the genome, Ihry, et al.,1 used one of SBI’s AAVS1 Safe Harbor Site Homologous Recombination (HR) Targeting Vectors. These HR Targeting Vectors are a great way to get reliable expression from a gene knock-in as they target your gene-of-interest to the AAVS1 site for consistent and robust gene expression (Figure 1). Knock-ins at the AAVS1 site show no phenotypic effects, and the DNA at this location is kept in an open conformation for reliable and consistent gene expression.
Learn more about SBI’s different AAVS1 Safe Harbor Targeting products here or see the many options for targeting other sites in the genome using any of our HR Targeting Vectors. We also offer a very robust, titratable inducible gene expression system similar to the one used by Ihry, et al.,1 but inducible with cumate—learn more here.
Figure 1:
REFERENCE
- Ihry RJ, et al. p53 inhibits CRISPR–Cas9 engineering in human pluripotent stem cells. Nature Medicine. 11 June 2018. 24:939–946.