Cas9 RT-PCR Primer Set
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| CAS9-PR-1 | Cas9 RT-PCR Primer Set (50 µL at 5 µM for each primer set) | 50 Reactions | $103 |
|
||||
Overview
Overview
Confirm Cas9 mRNA expression
With our ready-to-go Cas9 RT-PCR Primer Set, you can quickly and easily confirm Cas9 expression in cells or in vitro transcription assays.
The amplicon for Primer Set 1 is 219bp and amplicon for Primer Set 2 is 122bp. These primer sets allow for detection of Cas9 nuclease, Cas9 Nickase and Cas9 double mutant at messenger level. The primer sets are compatible with Cas9 mRNA expression from any SBI Cas9 SmartNuclease construct and vectors from Dr. Feng Zhang's lab as well.
How It Works
How It Works
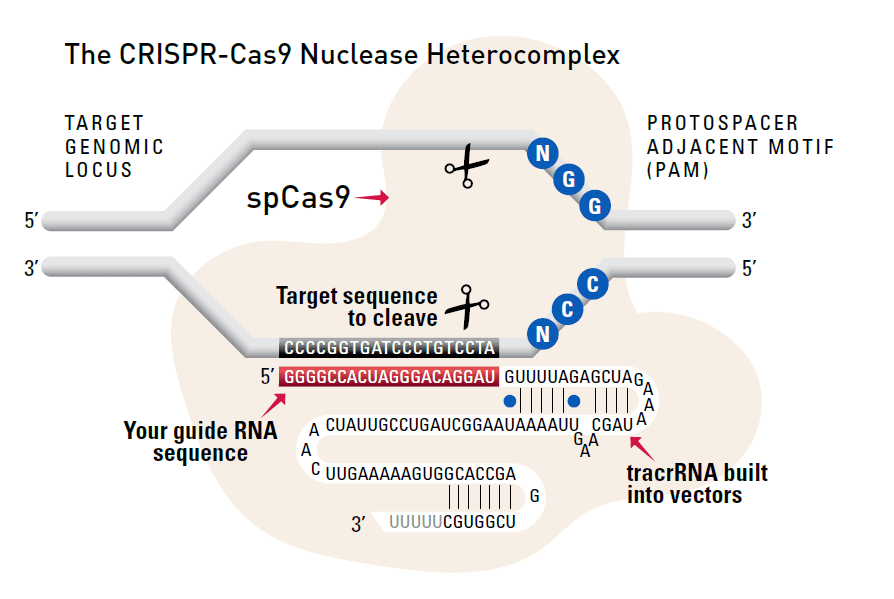
CRISPR/Cas9 Basics
Through careful selection of the target sequence and design of a donor plasmid for homologous recombination, you can achieve efficient and highly targeted genomic modification with CRISPR/Cas9.
The system
Cas9 protein—uses guide RNA (gRNA) to direct site-specific, double-strand DNA cleavage adjacent to a protospacer adapter motif (PAM) in the target DNA.
gRNA—RNA sequence that guides Cas9 to cleave a homologous region in the target genome. Efficient cleavage only where the gRNA homology is adjacent to a PAM.
PAM—protospacer adapter motif, NGG, is a target DNA sequence that spCas9 will cut upstream from if directed to by the gRNA.
The workflow at-a-glance
DESIGN: Select gRNA and HR donor plasmids. Choice of gRNA site and design of donor plasmid determines whether the homologous recombination event results in a knock-out, knock-in, edit, or tagging.
CONSTRUCT: Clone gRNA into all-in-one Cas9 vector. Clone 5’ and 3’ homology arms into HR donor plasmid. If creating a knock-in, clone desired gene into HR donor.
CO-TRANSFECT or CO-INJECT: Introduce Cas9, gRNA, and HR Donors into the target cells using co-transfection for plasmids, co-transduction for lentivirus, or co-injection for mRNAs.
SELECT/SCREEN: Select or screen for mutants and verify.
VALIDATE: Genotype or sequence putative mutants to verify single or biallelic conversion.
Supporting Data
Supporting Data
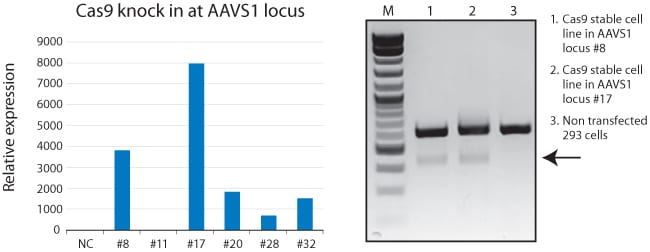
See our Cas9 RT-PCR Primer Set evaluate expression of Cas9 knocked-into the AAVS1 site
Figure 1. Use of the Cas9 RT-PCR Primer set to evaluate Cas9 expression after Cas9 knock-in at the AAVS1 site. We knocked the CAS9 gene into the AAVS1 site using the DIY Cas9-expressing Cells HR Donor and the All-in-one Cas9 SmartNuclease & AAVS1 gRNA Plasmid. We then tested the resulting positive clones for relative Cas9 mRNA expression using qPCR (left panel) and analyzed the two highest-expressing clones (#8 and 17) for Cas9-mediated cleavage activity using a Surveyor Assay (right panel, arrow indicated band showing Cas9-mediated cleavage). In the Surveyor Assay, only the two lanes with the Cas9 clones showed Cas9 activity (arrow), with the non-transfected cells showing no Cas9 activity.
FAQs
Resources
Related Products
Citations
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| CAS9-PR-1 | Cas9 RT-PCR Primer Set (50 µL at 5 µM for each primer set) | 50 Reactions | $103 |
|
||||
Overview
Overview
Confirm Cas9 mRNA expression
With our ready-to-go Cas9 RT-PCR Primer Set, you can quickly and easily confirm Cas9 expression in cells or in vitro transcription assays.
The amplicon for Primer Set 1 is 219bp and amplicon for Primer Set 2 is 122bp. These primer sets allow for detection of Cas9 nuclease, Cas9 Nickase and Cas9 double mutant at messenger level. The primer sets are compatible with Cas9 mRNA expression from any SBI Cas9 SmartNuclease construct and vectors from Dr. Feng Zhang's lab as well.
How It Works
How It Works
CRISPR/Cas9 Basics
Through careful selection of the target sequence and design of a donor plasmid for homologous recombination, you can achieve efficient and highly targeted genomic modification with CRISPR/Cas9.
The system
Cas9 protein—uses guide RNA (gRNA) to direct site-specific, double-strand DNA cleavage adjacent to a protospacer adapter motif (PAM) in the target DNA.
gRNA—RNA sequence that guides Cas9 to cleave a homologous region in the target genome. Efficient cleavage only where the gRNA homology is adjacent to a PAM.
PAM—protospacer adapter motif, NGG, is a target DNA sequence that spCas9 will cut upstream from if directed to by the gRNA.
The workflow at-a-glance
DESIGN: Select gRNA and HR donor plasmids. Choice of gRNA site and design of donor plasmid determines whether the homologous recombination event results in a knock-out, knock-in, edit, or tagging.
CONSTRUCT: Clone gRNA into all-in-one Cas9 vector. Clone 5’ and 3’ homology arms into HR donor plasmid. If creating a knock-in, clone desired gene into HR donor.
CO-TRANSFECT or CO-INJECT: Introduce Cas9, gRNA, and HR Donors into the target cells using co-transfection for plasmids, co-transduction for lentivirus, or co-injection for mRNAs.
SELECT/SCREEN: Select or screen for mutants and verify.
VALIDATE: Genotype or sequence putative mutants to verify single or biallelic conversion.
Supporting Data
Supporting Data
See our Cas9 RT-PCR Primer Set evaluate expression of Cas9 knocked-into the AAVS1 site
Figure 1. Use of the Cas9 RT-PCR Primer set to evaluate Cas9 expression after Cas9 knock-in at the AAVS1 site. We knocked the CAS9 gene into the AAVS1 site using the DIY Cas9-expressing Cells HR Donor and the All-in-one Cas9 SmartNuclease & AAVS1 gRNA Plasmid. We then tested the resulting positive clones for relative Cas9 mRNA expression using qPCR (left panel) and analyzed the two highest-expressing clones (#8 and 17) for Cas9-mediated cleavage activity using a Surveyor Assay (right panel, arrow indicated band showing Cas9-mediated cleavage). In the Surveyor Assay, only the two lanes with the Cas9 clones showed Cas9 activity (arrow), with the non-transfected cells showing no Cas9 activity.