NANOPARTICLE TRACKING ANALYSIS (NTA) EXOSOME CHARACTERIZATION
Know what’s in your sample—choose conventional or fluorescent NTA with SBI’s NTA Service and get quality data on exosome particle size and concentration.
Get the confidence to move forward with SBI’s NTA Exosome Services
Why choose SBI for NTA characterization of exosome preps?
Because we've been working with exosomes for years, we can reliably and reproducibly isolate exosomes from almost any biofluid including:
* Plasma
* Tissue culture media
* CSF
* Synovial Fluid.
We also have the ability to handle challenging samples, such as mouse bronchial alveolar fluid.
And because SBI is the only company that can provide fluorescent NTA using our proprietary ExoGlow™-NTA reagents.
What’s NTA?
SBI uses NTA measurements that rely on light scattering to extract particle size and the number of particles in a sample (Figure 1A). Briefly, a laser is directed at the sample and the particles in the sample scatter the light, which is imaged by a camera attached to a microscope and operating at 30 frames per second. The NTA software collects data on multiple particles simultaneously, and calculates the hydrodynamic diameter of each particle using the Stokes-Einstein equation.
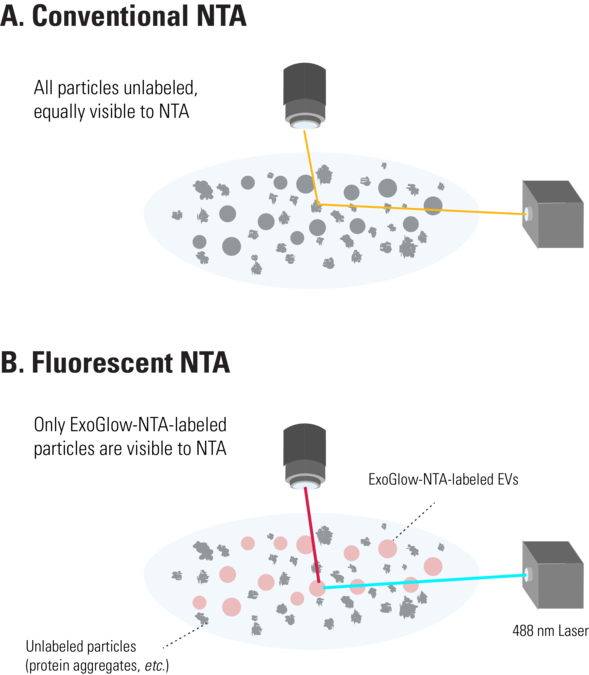
Figure 1. The ExoGlow-NTA dye only binds to membranes of intact EVs. Unlike conventional NTA (A), which collects data on all particles in a solution based on light scattering, the fluorescence mode of the NTA instrument selectively detects only the fluorescently-labeled particles—in this case, the ExoGlow-NTA-labeled EVs (B)—and only the data from fluorescently-labeled particles is reported.
What is fluorescent NTA and how is it different from conventional NTA?
Making a great exosome NTA research tool even better, SBI has developed ExoGlow-NTA, a proprietary dye for fluorescent NTA* that enables data collection for only the extracellular vesicles present in a heterogenous sample. The result is more accurate EV/exosomal NanoSight data that ignores protein aggregates, membrane fractions, and other background particles to provide EV-specific particle size and concentration.
The ExoGlow-NTA reagent works by binding specifically and efficiently to the membranes of intact EVs, including exosomes. During data collection for fluorescent NTA, only the light scattering from fluorescently-labeled particles is obtained, resulting in EV-specific signal that ignores membrane fragments, protein aggregates, and other background particles since they do not bind the ExoGlow-NTA dye (Figure 1B). Thus, with the ExoGlow-NTA Kit, the data delivered by the NTA analysis more accurately reports on the EVs in your sample, rather than all of the particles in your sample.
Nanoparticle Tracking Analysis (NTA) Exosome Characterization Services highlights:
Understand the quality of your exosome preps
Leverage SBI’s long-term experience working with exosomes
Send us your biofluid or exosome prep and receive data in about 2 weeks
How SBI's NTA Exosome Characterization Services Work
Supporting Data
Get Better NTA data on EVs using ExoGlow-NTA
SBI’s ExoGlow-NTA Fluorescent Labeling Kit delivers highly accurate EV quantitation in a quick and easy workflow. The proprietary dye offers highly efficient labeling (Figure 2), very low background (Figure 3), and is compatible with a wide range of EV isolation techniques (Figure 4).
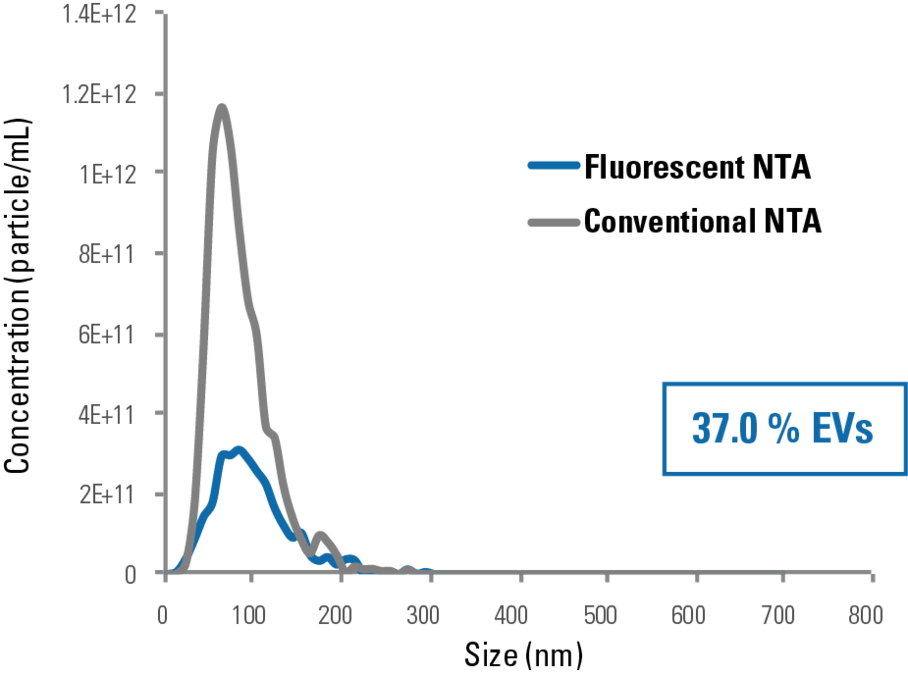
Figure 2. ExoGlow-NTA-labeled liposomes deliver consistent NTA data whether in light scattering or fluorescent mode. The high concordance of NTA and fluorescent NTA data collected from the ExoGlow-NTA Kit internal standards (ExoGlow-NTA-labeled synthetic liposomes) demonstrates the labeling efficiency of the ExoGlow-NTA Dye and accuracy of the fluorescent NTA method for characterizing EVs.
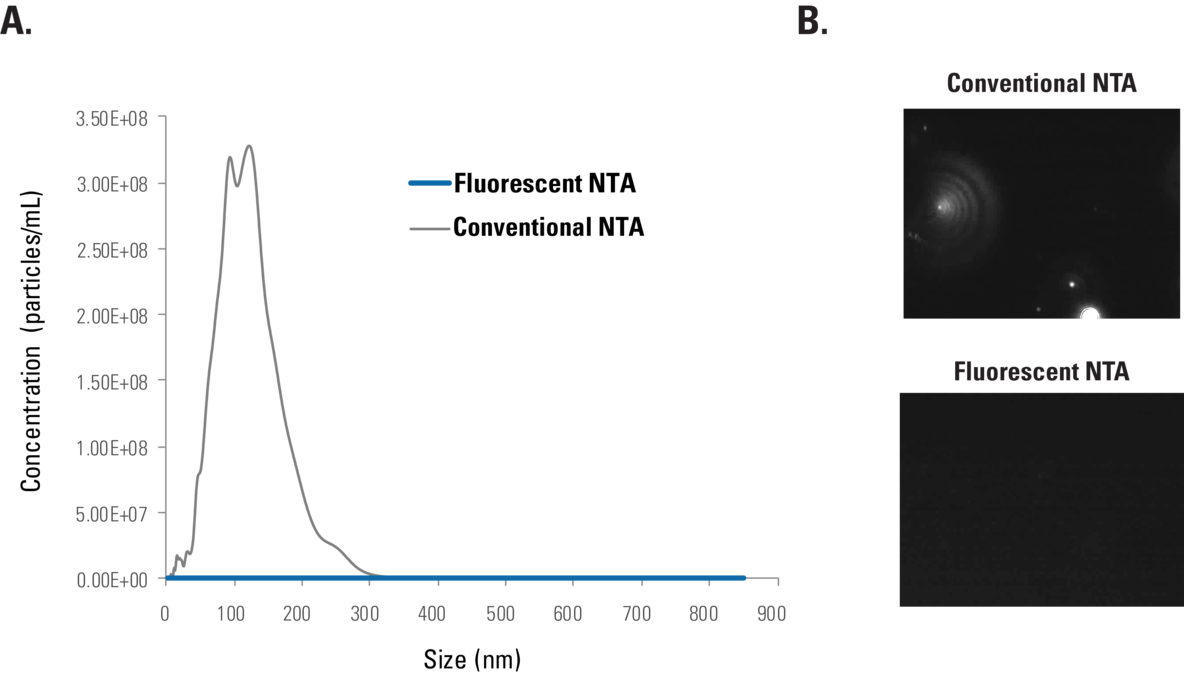
Figure 3.ExoGlow-NTA exhibits undetectable background signal. Conventional NTA and Fluorescent NTA of ExoGlow-NTA dye in the absence of EVs shows bias-free undetectable autofluorescence, based on (A) particle counts and (B) imaging.
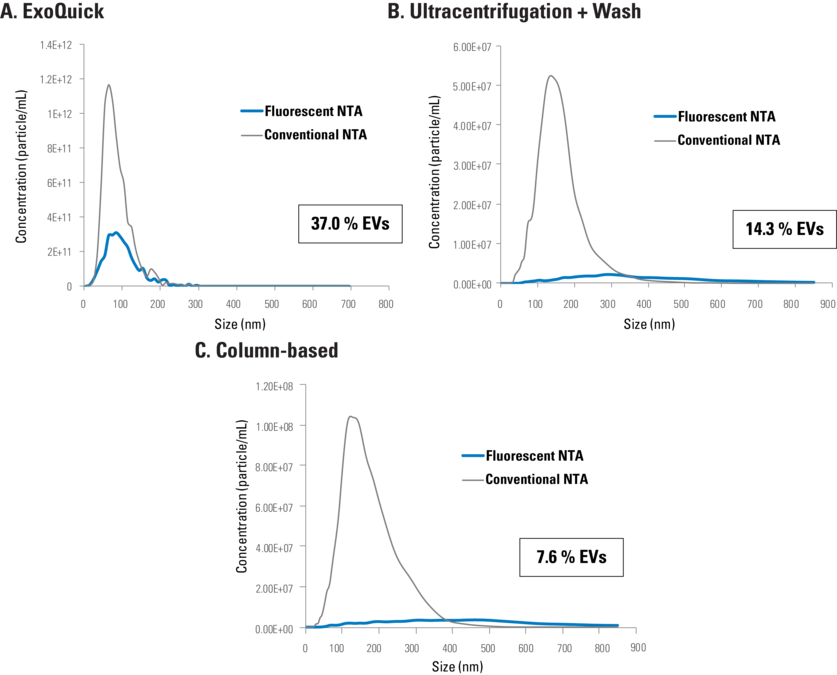
Figure 4. ExoGlow-NTA demonstrates that conventional NTA overestimates EV concentration in samples irrespective of EV isolation method. Representative data comparing conventional NTA and fluorescent NTA for EVs isolated using (A) ExoQuick (10 μg serum protein), (B) ultracentrifugation and wash (1 μg serum protein), or (C) column based (1 μg serum protein), shows how much of the conventional NTA signal is due to non-EV particles.


