ExoQuick®-TC UltraPure
Scalable EV Isolation. Maximum Purity. No Ultracentrifuge.
Why should scale come at the cost of purity?
With SBI's ExoQuick®-TC UltraPure, you get both. This dual-action EV isolation kit combines SBI's trusted ExoQuick® precipitation with advanced mixed-mode affinity purification to deliver maximum yields of pure, lipoprotein-free, intact EVs in under 20 minutes of hands-on time — no ultracentrifuge, no pre-concentration, no ultrafiltration required. Process 10 mL to 1 L of conditioned media in a single reaction, whether from a standard tissue culture flask or a large-scale bioreactor harvest.
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| EQ-TC UltraPure_S | ExoQuick®-TC UltraPure EV Isolation Kit for Tissue Culture *1 reaction = 10 mL Culture Medium |
10 Reactions | $799 |
|
||||
| EQ-TC UltraPure_L | ExoQuick®-TC UltraPure EV Isolation Kit for Tissue Culture *1 reaction = 100 mL Culture Medium |
10 Reactions | $3999 |
|
||||
Kit Contents (Shipped at Ambient Temperature)
| Component | Volume / Quantity | Storage Temperature | |
|---|---|---|---|
| EQ-TC UltraPure_S | ExoQuick® | 10 mL, Sterile | +4°C to +30°C |
| UltraPure Purification Column (Small) | 10 columns | ||
| Column Buffer | 25 mL | ||
| Collection Tubes | 10 | ||
| 2 ml Eppendorf Tubes | 10 | ||
| EQ-TC UltraPure_L | ExoQuick® | 200 mL, Sterile | +4°C to +30°C |
| UltraPure Purification Column (Large) | 10 columns | ||
| Column Buffer | 150 mL |
Note: In figures and tables, this product may be referred to as EQ-TC UltraPure (its catalog reference) for space efficiency. Both names refer to the same product and can be used interchangeably.
Overview
Overview
ExoQuick®-TC UltraPure is engineered for researchers who demand high-purity, lipoprotein-free extracellular vesicles from tissue culture media - at any scale. This advanced kit incorporates SBI's trusted ExoQuick® EV precipitation reagent with innovative multimodal chromatography resin, which has dual‑functionality: size separation and binding chromatography. The ligand within the core bead combines hydrophobic interactions with both positive and negative ionic functionalities, enabling it to bind impurities through hydrophobic interactions and electrostatic attraction. This mechanism facilitates the removal of impurities such as DNA and carryover proteins, including lipoproteins. This dual-action workflow ensures:
- All EVs are captured by precipitation using ExoQuick® reagents, achieving >90% EV recovery from conditioned media
- Maximum purity through mixed-mode chromatography that integrates size exclusion, affinity interactions, and lipoprotein depletion — removing albumin (BSA), IgG, and lipoproteins in a single pass
Key Benefits of ExoQuick®-TC UltraPure
- >90% EV Recovery — dual-action precipitation and column purification ensures maximum EV capture from every reaction
- Near-Complete Lipoprotein Depletion — ApoB ELISA confirms near-undetectable lipoprotein levels, eliminating the most common confounder in EV RNA analysis and sequencing
- Scalable from 10 mL to 1 L — process standard flask volumes or large bioreactor harvests in a single reaction; no pre-concentration, no ultrafiltration, no pooling
- Minimal Albumin and IgG Carry-Over — Western blot and Ponceau S staining confirm cleaner preparations than SEC and TFF
- Consistent and Reproducible — unlike ultracentrifugation, ExoQuick®-TC UltraPure delivers highly reproducible results across experiments without specialized equipment
- Fast, Simple Workflow — add ExoQuick® to conditioned media, mix, centrifuge, transfer to column, brief incubation, quick spin — done in under 20 minutes of hands-on time
Downstream Applications
ExoQuick®-TC UltraPure generates EVs ready for a full range of downstream applications, including:
- EV quantification and characterisation (NTA, TEM, Western blot, ELISA)
- Clean EV preparation for NGS (next-generation sequencing), small RNA sequencing, and miRNA profiling
- RNA extraction and protein profiling
- Mass spectrometry proteomics (in-gel digestion compatible)
- EV engineering and cargo loading (compatible with XPACK)
- EV uptake assays and cargo delivery (e.g., copGFP-loaded EVs)
- In vivo / ex vivo EV delivery and administration for in vivo studies
- EV biomarker discovery and therapeutic EV development
With its scalability, simplicity, and superior performance, ExoQuick®-TC UltraPure is the practical choice for tissue culture EV workflows — from small-scale discovery to large-scale bioreactor production.
References
How It Works
How It Works
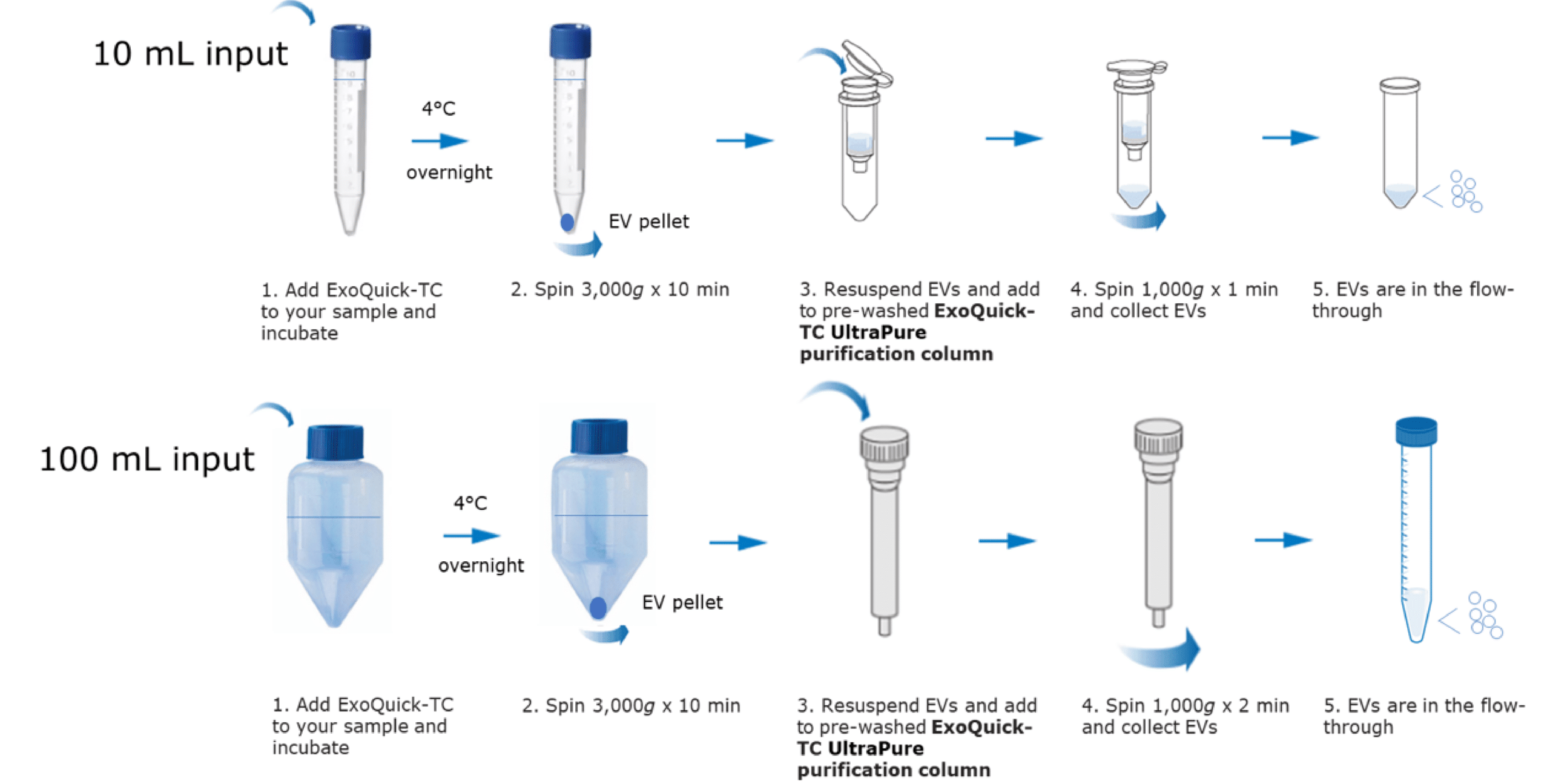
ExoQuick®-TC UltraPure uses a dual-action workflow: ExoQuick® precipitation captures >90% of EVs, followed by a mixed-mode affinity column that removes albumin, IgG, and lipoproteins in a single pass. No ultracentrifuge. No pre-concentration. Under 20 minutes of hands-on time.
Figure 1. The simple ExoQuick®-TC UltraPure workflow ExoQuick reagent captures virtually all EVs in a sample, achieving over 90% EV recovery. This is followed by a specialized chromatography column that enhances separation by combining size-based and chemical-property-based mechanisms, removing BSA, IgG, and lipoproteins.
Simple 5-Step Workflow (10 mL to 1 L compatible):
- Step 1 — Add ExoQuick® to conditioned media
- Step 2 — Mix and incubate at 4°C
- Step 3 — Centrifuge to pellet EVs and discard supernatant
- Step 4 — Resuspend and load onto the purification column
- Step 5 — Collect purified EVs from the flow-through
Total hands-on time: <20 minutes. No ultracentrifuge. No pre-concentration. No specialized equipment.
Supporting Data
Supporting Data
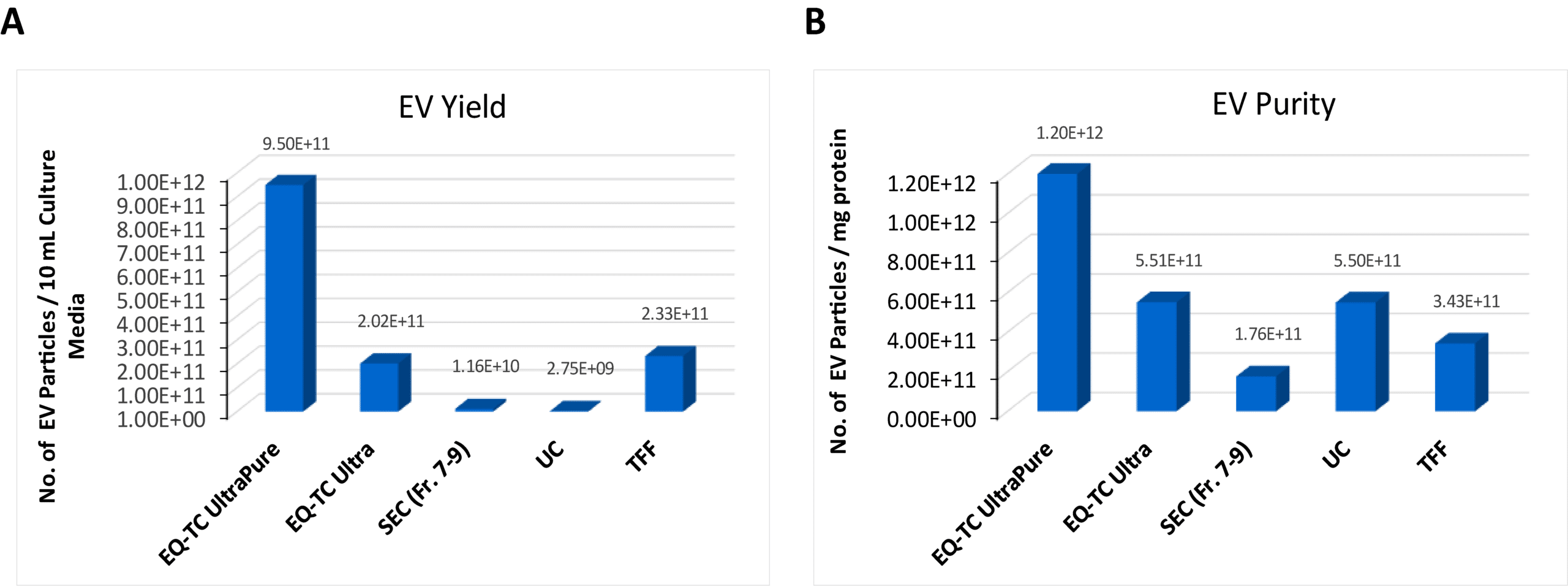
ExoQuick®-TC UltraPure Delivers >50× Higher EV Yield vs. SEC
Figure 2. ExoQuick®-TC UltraPure provides higher EV yield and purity compared to other isolation methods. EVs were isolated from HEK293 cell culture medium using ExoQuick®‑TC Ultra, ExoQuick®‑TC UltraPure, size‑exclusion chromatography (SEC)–based qEV, ultracentrifugation, and tangential flow filtration (TFF). EV particle numbers from each method were quantified by NTA. NTA confirms that ExoQuick®‑TC UltraPure delivers superior EV yield and purity, as shown by both the volume of input culture medium used per 10 mL (A) and the input protein amount per mg (B). EV protein levels were quantified using the Qubit assay.
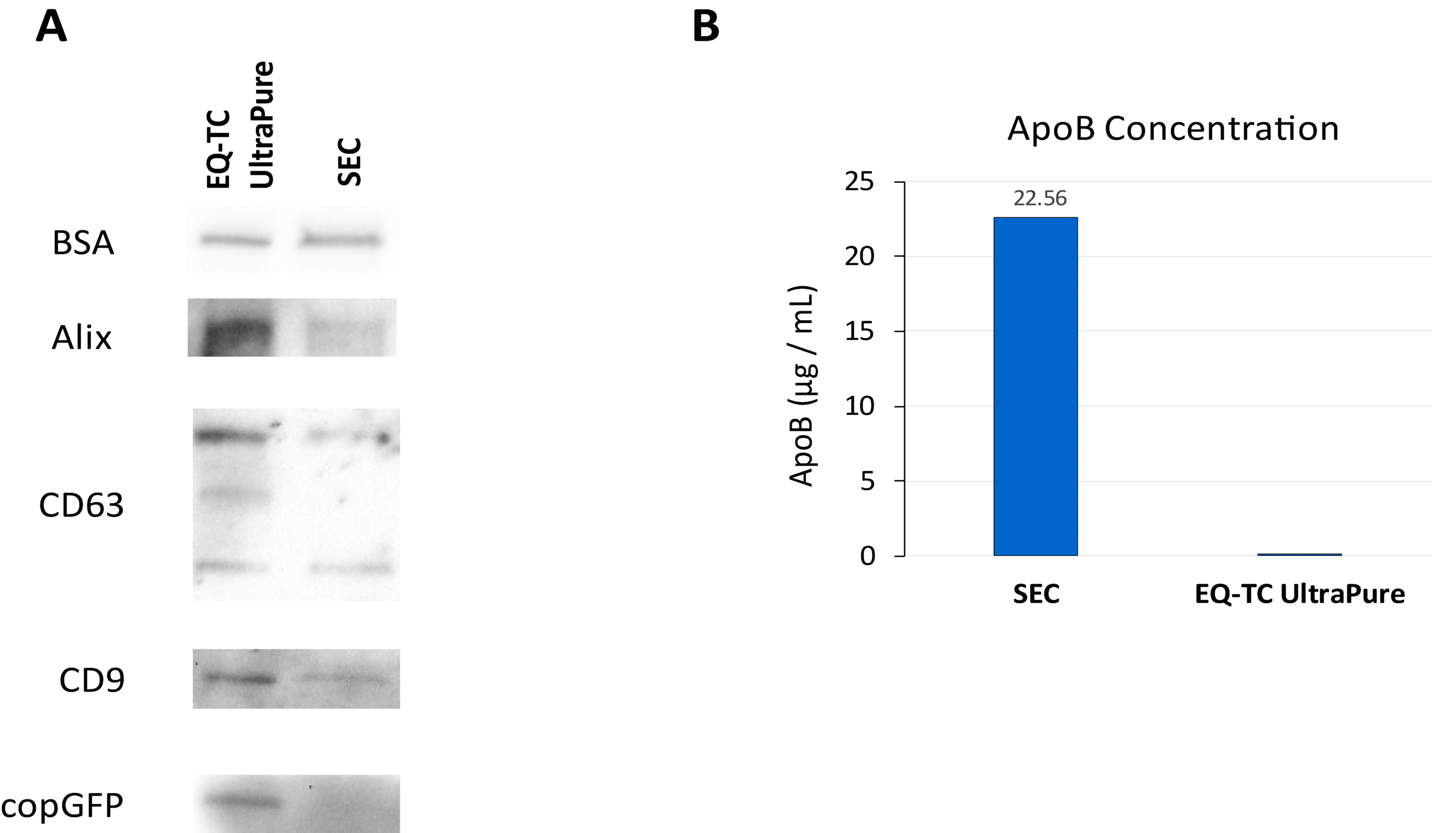
EQ-TC UltraPure = Cleaner EVs, Near-Complete Lipoprotein Removal
Figure 3. ExoQuick®-TC UltraPure delivers higher yield and superior purity compared to SEC-based qEV. Culture medium (10 mL) from the XPACK530 HEK293 stable cell line – containing copGFP-loaded EVs – was used for isolation. (A) Western blot analysis shows that EVs isolated with ExoQuick®-TC UltraPure contain higher levels of EV-specific markers and exhibit minimal albumin carryover compared to the SEC-based qEV method, indicating both higher yield and superior purity. EV integrity is also well preserved, as indicated by robust copGFP expression. (B) ApoB ELISA analysis reveals that EVs isolated using ExoQuick®-TC UltraPure contain nearly undetectable ApoB levels, confirming minimal lipoprotein contamination.
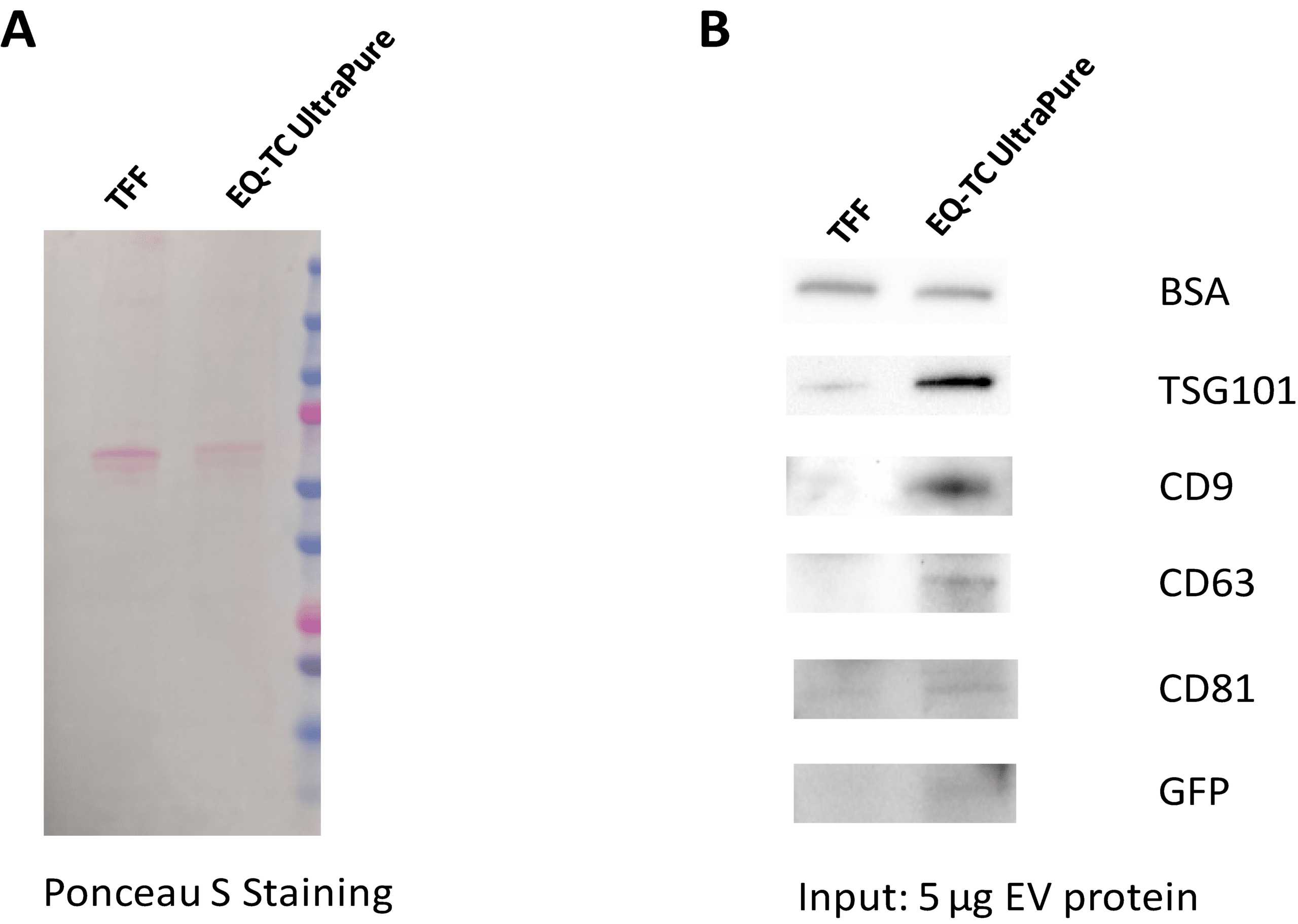
Cleaner Large-Volume EV Preps Than TFF — at 100 mL Scale
Figure 4. ExoQuick®-TC UltraPure is suitable for large-scale EV isolation while maintaining superior quality. For this experiment, 100 mL of culture medium from an XPACK530 stable cell line - containing copGFP-loaded EVs - was processed as one reaction. (A) Ponceau S staining confirms that ExoQuick®-TC UltraPure preparations are significantly cleaner compared to tangential flow filtration (TFF) (B) Western blot analysis demonstrates higher EV yield, strong EV marker expression, greater purity with minimal albumin carryover, and improved vesicle integrity indicated by copGFP expression, all compared to TFF.
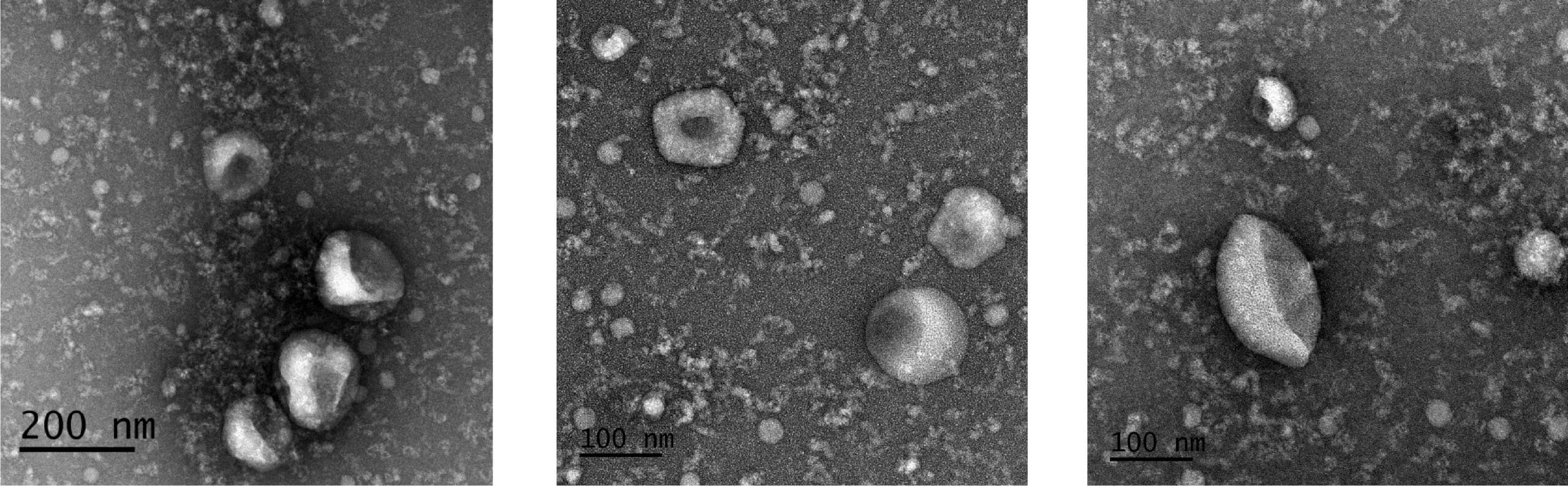
TEM Confirms Native EV Morphology Is Preserved
Figure 5. EVs isolated using ExoQuick®-TC UltraPure display typical native EV morphology. Transmission electron micrographs of EVs isolated from HEK 293 cell culture medium using ExoQuick®-TC UltraPure. The same sample is shown at two different magnifications. Multiple vesicles with typical EV morphology are visible in each image.
FAQs
ExoQuick®-TC UltraPure outperforms both SEC (e.g., qEV) and tangential flow filtration (TFF) in yield and purity for cell culture media applications.
vs. SEC (qEV): Delivers more than 50× higher EV yield, with superior removal of albumin, IgG, and lipoproteins. SEC requires pre-concentration of conditioned media before loading — ExoQuick®-TC UltraPure processes your sample directly, no pre-concentration required.
vs. TFF: At equivalent large-volume inputs (100 mL), EQ-TC UltraPure produces significantly cleaner EV preparations — confirmed by Ponceau S staining and Western blot — with higher EV marker expression and lower protein contamination. Unlike TFF, no specialised tangential flow filtration system is required.
EVs isolated with ExoQuick®-TC UltraPure are validated for:
- Nanoparticle Tracking Analysis (NTA)
- Transmission Electron Microscopy (TEM)
- Western blot (EV marker detection)
- ELISA
- RNA extraction, small RNA/miRNA sequencing, and next-generation sequencing (NGS) — compatible with SBI's NGS service
- Mass spectrometry proteomics (in-gel digestion)
- EV uptake and cargo delivery assays
- In vivo / ex vivo administration studies
Documentation
Citations
Related Products
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| EQ-TC UltraPure_S | ExoQuick®-TC UltraPure EV Isolation Kit for Tissue Culture *1 reaction = 10 mL Culture Medium |
10 Reactions | $799 |
|
||||
| EQ-TC UltraPure_L | ExoQuick®-TC UltraPure EV Isolation Kit for Tissue Culture *1 reaction = 100 mL Culture Medium |
10 Reactions | $3999 |
|
||||
Kit Contents (Shipped at Ambient Temperature)
| Component | Volume / Quantity | Storage Temperature | |
|---|---|---|---|
| EQ-TC UltraPure_S | ExoQuick® | 10 mL, Sterile | +4°C to +30°C |
| UltraPure Purification Column (Small) | 10 columns | ||
| Column Buffer | 25 mL | ||
| Collection Tubes | 10 | ||
| 2 ml Eppendorf Tubes | 10 | ||
| EQ-TC UltraPure_L | ExoQuick® | 200 mL, Sterile | +4°C to +30°C |
| UltraPure Purification Column (Large) | 10 columns | ||
| Column Buffer | 150 mL |
Note: In figures and tables, this product may be referred to as EQ-TC UltraPure (its catalog reference) for space efficiency. Both names refer to the same product and can be used interchangeably.
Overview
Overview
ExoQuick®-TC UltraPure is engineered for researchers who demand high-purity, lipoprotein-free extracellular vesicles from tissue culture media - at any scale. This advanced kit incorporates SBI's trusted ExoQuick® EV precipitation reagent with innovative multimodal chromatography resin, which has dual‑functionality: size separation and binding chromatography. The ligand within the core bead combines hydrophobic interactions with both positive and negative ionic functionalities, enabling it to bind impurities through hydrophobic interactions and electrostatic attraction. This mechanism facilitates the removal of impurities such as DNA and carryover proteins, including lipoproteins. This dual-action workflow ensures:
- All EVs are captured by precipitation using ExoQuick® reagents, achieving >90% EV recovery from conditioned media
- Maximum purity through mixed-mode chromatography that integrates size exclusion, affinity interactions, and lipoprotein depletion — removing albumin (BSA), IgG, and lipoproteins in a single pass
Key Benefits of ExoQuick®-TC UltraPure
- >90% EV Recovery — dual-action precipitation and column purification ensures maximum EV capture from every reaction
- Near-Complete Lipoprotein Depletion — ApoB ELISA confirms near-undetectable lipoprotein levels, eliminating the most common confounder in EV RNA analysis and sequencing
- Scalable from 10 mL to 1 L — process standard flask volumes or large bioreactor harvests in a single reaction; no pre-concentration, no ultrafiltration, no pooling
- Minimal Albumin and IgG Carry-Over — Western blot and Ponceau S staining confirm cleaner preparations than SEC and TFF
- Consistent and Reproducible — unlike ultracentrifugation, ExoQuick®-TC UltraPure delivers highly reproducible results across experiments without specialized equipment
- Fast, Simple Workflow — add ExoQuick® to conditioned media, mix, centrifuge, transfer to column, brief incubation, quick spin — done in under 20 minutes of hands-on time
Downstream Applications
ExoQuick®-TC UltraPure generates EVs ready for a full range of downstream applications, including:
- EV quantification and characterisation (NTA, TEM, Western blot, ELISA)
- Clean EV preparation for NGS (next-generation sequencing), small RNA sequencing, and miRNA profiling
- RNA extraction and protein profiling
- Mass spectrometry proteomics (in-gel digestion compatible)
- EV engineering and cargo loading (compatible with XPACK)
- EV uptake assays and cargo delivery (e.g., copGFP-loaded EVs)
- In vivo / ex vivo EV delivery and administration for in vivo studies
- EV biomarker discovery and therapeutic EV development
With its scalability, simplicity, and superior performance, ExoQuick®-TC UltraPure is the practical choice for tissue culture EV workflows — from small-scale discovery to large-scale bioreactor production.
References
How It Works
How It Works
ExoQuick®-TC UltraPure uses a dual-action workflow: ExoQuick® precipitation captures >90% of EVs, followed by a mixed-mode affinity column that removes albumin, IgG, and lipoproteins in a single pass. No ultracentrifuge. No pre-concentration. Under 20 minutes of hands-on time.
Figure 1. The simple ExoQuick®-TC UltraPure workflow ExoQuick reagent captures virtually all EVs in a sample, achieving over 90% EV recovery. This is followed by a specialized chromatography column that enhances separation by combining size-based and chemical-property-based mechanisms, removing BSA, IgG, and lipoproteins.
Simple 5-Step Workflow (10 mL to 1 L compatible):
- Step 1 — Add ExoQuick® to conditioned media
- Step 2 — Mix and incubate at 4°C
- Step 3 — Centrifuge to pellet EVs and discard supernatant
- Step 4 — Resuspend and load onto the purification column
- Step 5 — Collect purified EVs from the flow-through
Total hands-on time: <20 minutes. No ultracentrifuge. No pre-concentration. No specialized equipment.
Supporting Data
Supporting Data
ExoQuick®-TC UltraPure Delivers >50× Higher EV Yield vs. SEC
Figure 2. ExoQuick®-TC UltraPure provides higher EV yield and purity compared to other isolation methods. EVs were isolated from HEK293 cell culture medium using ExoQuick®‑TC Ultra, ExoQuick®‑TC UltraPure, size‑exclusion chromatography (SEC)–based qEV, ultracentrifugation, and tangential flow filtration (TFF). EV particle numbers from each method were quantified by NTA. NTA confirms that ExoQuick®‑TC UltraPure delivers superior EV yield and purity, as shown by both the volume of input culture medium used per 10 mL (A) and the input protein amount per mg (B). EV protein levels were quantified using the Qubit assay.
EQ-TC UltraPure = Cleaner EVs, Near-Complete Lipoprotein Removal
Figure 3. ExoQuick®-TC UltraPure delivers higher yield and superior purity compared to SEC-based qEV. Culture medium (10 mL) from the XPACK530 HEK293 stable cell line – containing copGFP-loaded EVs – was used for isolation. (A) Western blot analysis shows that EVs isolated with ExoQuick®-TC UltraPure contain higher levels of EV-specific markers and exhibit minimal albumin carryover compared to the SEC-based qEV method, indicating both higher yield and superior purity. EV integrity is also well preserved, as indicated by robust copGFP expression. (B) ApoB ELISA analysis reveals that EVs isolated using ExoQuick®-TC UltraPure contain nearly undetectable ApoB levels, confirming minimal lipoprotein contamination.
Cleaner Large-Volume EV Preps Than TFF — at 100 mL Scale
Figure 4. ExoQuick®-TC UltraPure is suitable for large-scale EV isolation while maintaining superior quality. For this experiment, 100 mL of culture medium from an XPACK530 stable cell line - containing copGFP-loaded EVs - was processed as one reaction. (A) Ponceau S staining confirms that ExoQuick®-TC UltraPure preparations are significantly cleaner compared to tangential flow filtration (TFF) (B) Western blot analysis demonstrates higher EV yield, strong EV marker expression, greater purity with minimal albumin carryover, and improved vesicle integrity indicated by copGFP expression, all compared to TFF.
TEM Confirms Native EV Morphology Is Preserved
Figure 5. EVs isolated using ExoQuick®-TC UltraPure display typical native EV morphology. Transmission electron micrographs of EVs isolated from HEK 293 cell culture medium using ExoQuick®-TC UltraPure. The same sample is shown at two different magnifications. Multiple vesicles with typical EV morphology are visible in each image.
FAQs
ExoQuick®-TC UltraPure outperforms both SEC (e.g., qEV) and tangential flow filtration (TFF) in yield and purity for cell culture media applications.
vs. SEC (qEV): Delivers more than 50× higher EV yield, with superior removal of albumin, IgG, and lipoproteins. SEC requires pre-concentration of conditioned media before loading — ExoQuick®-TC UltraPure processes your sample directly, no pre-concentration required.
vs. TFF: At equivalent large-volume inputs (100 mL), EQ-TC UltraPure produces significantly cleaner EV preparations — confirmed by Ponceau S staining and Western blot — with higher EV marker expression and lower protein contamination. Unlike TFF, no specialised tangential flow filtration system is required.
EVs isolated with ExoQuick®-TC UltraPure are validated for:
- Nanoparticle Tracking Analysis (NTA)
- Transmission Electron Microscopy (TEM)
- Western blot (EV marker detection)
- ELISA
- RNA extraction, small RNA/miRNA sequencing, and next-generation sequencing (NGS) — compatible with SBI's NGS service
- Mass spectrometry proteomics (in-gel digestion)
- EV uptake and cargo delivery assays
- In vivo / ex vivo administration studies