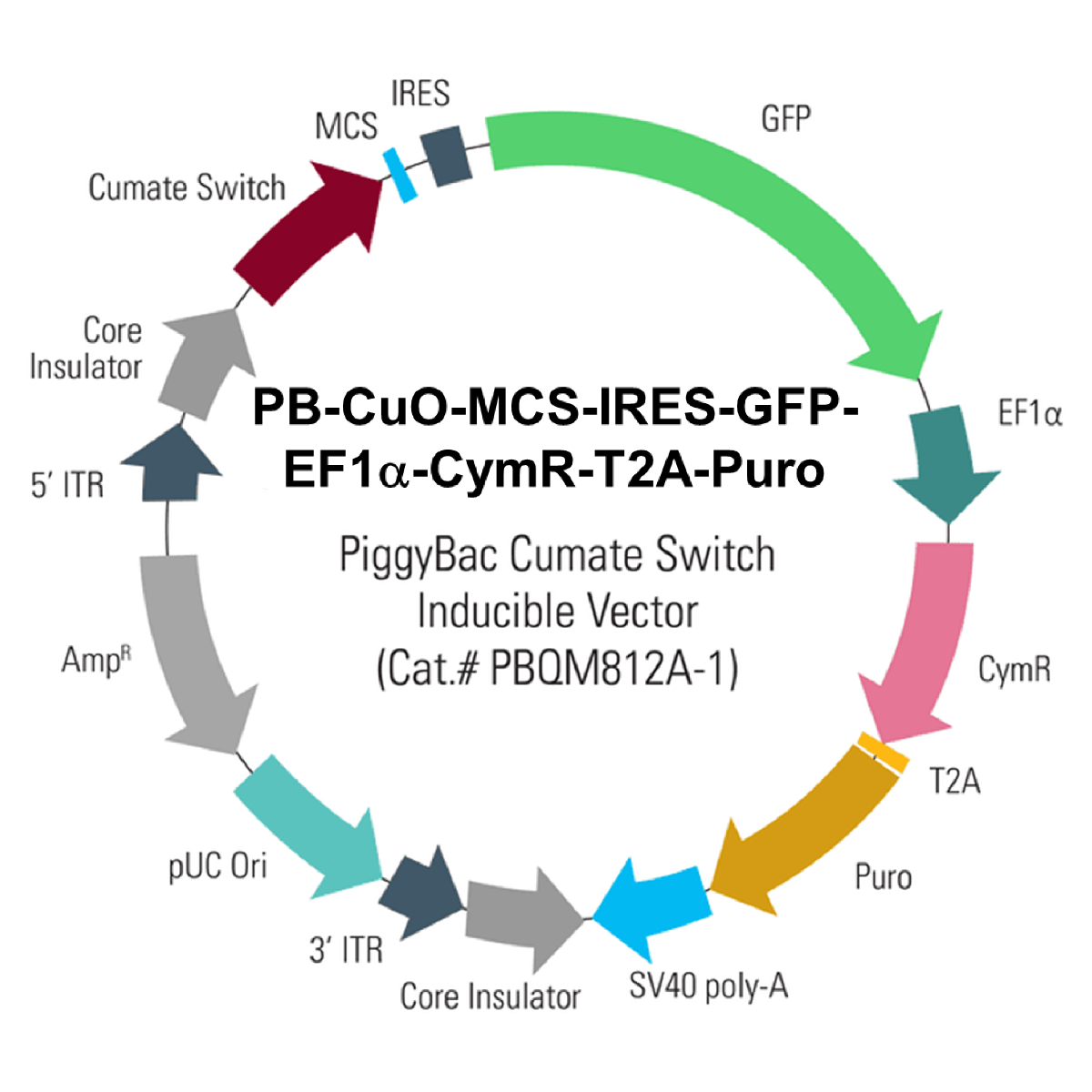
PB-Cuo-MCS-IRES-GFP-EF1α-CymR-T2A-Puro Inducible cDNA Cloning and Expression Vector
Combining easy PiggyBac transgenesis with our robust and titratable cumate-inducible expression system, this PiggyBac Vector is designed for cDNA expression
- Make transgenic cell lines with a single transfection
- Integrate multiple PiggyBac Vectors in a single transfection
- Insert an expression cassette into human, mouse, and rat cells
- Deliver virtually any-sized DNA insert, from 10 – 100 kb
- Choose from PiggyBac Vectors that express your gene-of-interest from constitutive or inducible promoters and include a variety of markers
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| PBQM812A-1 | PB-Cuo-MCS-IRES-GFP-EF1α-CymR-T2A-Puro Inducible cDNA Cloning and Expression Vector | 10 µg | $1620 |
|
||||
Overview
Overview
Robust, titratable gene expression delivered using the PiggyBac Transposon System
More than just easy, consistent transgenesis with PiggyBac, the PB-Cuo-MCS-IRES-GFP-EF1α-CymR-T2A-Puro Inducible cDNA Cloning and Expression Vector (Cat.# PBQM812A-1) adds in the robust and titratable gene expression control of SBI’s cumate-inducible expression system. Clone your gene-of-interest into the MCS for cumate-inducible expression, which you can quantitatively monitor with the co-expressed GFP. This vector also co-expresses the cumate repressor, CymR, and puromycin resistance from the EF1α promoter.
With the PiggyBac Transposon System, you can:
- Make transgenic cell lines with a single transfection
- Integrate multiple PiggyBac Vectors in a single transfection
- Insert an expression cassette into human, mouse, and rat cells
- Deliver virtually any-sized DNA insert, from 10 – 100 kb
- Choose from PiggyBac Vectors that express your gene-of-interest from constitutive or inducible promoters and include a variety of markers
- Determine the number of integration events with the PiggyBac qPCR Copy Number Kit (# PBC100A-1)
Customer Agreements
Academic customers can purchase PiggyBac Transposon System components for internal research purposes for indefinite use, whereas commercial customers must sign a customer agreement for a six-month, limited-use license to evaluate the technology.
* SBI is fully licensed to distribute PiggyBac vectors as a partnership with Hera BioLabs, Inc.
References
How It Works
How It Works
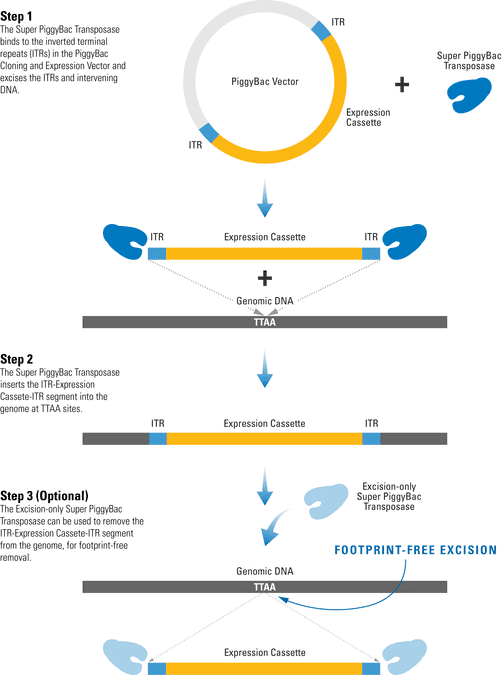
The PiggyBac Transposon System’s Cut-and-Paste Mechanism
The efficient PiggyBac Transposon System uses a cut-and-paste mechanism to transfer DNA from the PiggyBac Vector into the genome. If only temporary genomic integration is desired, the Excision-only PiggyBac Transposase can be transiently expressed for footprint-free removal of the insert, resulting in reconstitution of the original genome sequence.
Figure 1. The PiggyBac Transposon System’s cut-and-paste mechanism.
- The Super PiggyBac Transposase binds to specific inverted terminal repeats (ITRs) in the PiggyBac Cloning and Expression Vector and excises the ITRs and intervening DNA.
- The Super PiggyBac Transposase inserts the ITR-Expression Cassette-ITR segment into the genome at TTAA sites.
- The Excision-only Super PiggyBac Transposase can be used to remove the ITR-Expression Cassette-ITR segment from the genome, for footprint-free removal
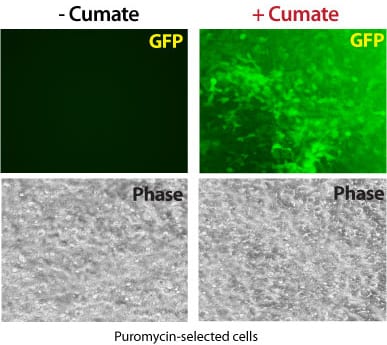
Tightly-controlled, inducible gene expression
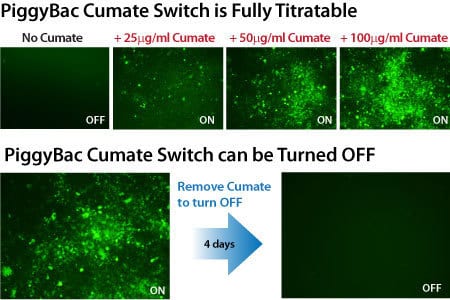
Get robust, titratable gene expression with low background using SBI’s cumate-inducible vectors. These vectors take advantage of CymR, a repressor that binds to cumate operator sequences (CuO) with high affinity in the absence of cumate, a non-toxic small molecule. Providing much lower background expression than similar systems, SBI’s cumate-inducible vectors can provide up to 32-fold induction of gene expression.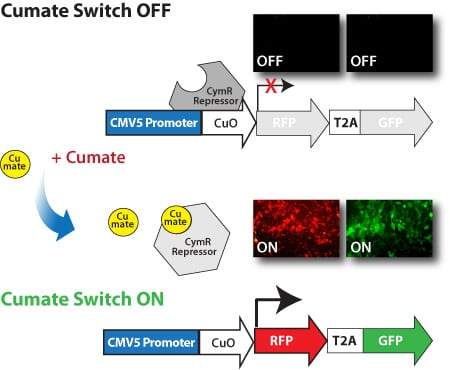
- Robust—increase expression up to 32-fold
- Adjustable—tune expression levels by titrating the amount of cumate
- Reversible—turn expression on, then off, then on again
- Powerful—suitable for in vivo applications
Supporting Data
FAQs
Documentation
Citations
Related Products
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| PBQM812A-1 | PB-Cuo-MCS-IRES-GFP-EF1α-CymR-T2A-Puro Inducible cDNA Cloning and Expression Vector | 10 µg | $1620 |
|
||||
Overview
Overview
Robust, titratable gene expression delivered using the PiggyBac Transposon System
More than just easy, consistent transgenesis with PiggyBac, the PB-Cuo-MCS-IRES-GFP-EF1α-CymR-T2A-Puro Inducible cDNA Cloning and Expression Vector (Cat.# PBQM812A-1) adds in the robust and titratable gene expression control of SBI’s cumate-inducible expression system. Clone your gene-of-interest into the MCS for cumate-inducible expression, which you can quantitatively monitor with the co-expressed GFP. This vector also co-expresses the cumate repressor, CymR, and puromycin resistance from the EF1α promoter.

With the PiggyBac Transposon System, you can:
- Make transgenic cell lines with a single transfection
- Integrate multiple PiggyBac Vectors in a single transfection
- Insert an expression cassette into human, mouse, and rat cells
- Deliver virtually any-sized DNA insert, from 10 – 100 kb
- Choose from PiggyBac Vectors that express your gene-of-interest from constitutive or inducible promoters and include a variety of markers
- Determine the number of integration events with the PiggyBac qPCR Copy Number Kit (# PBC100A-1)
Customer Agreements
Academic customers can purchase PiggyBac Transposon System components for internal research purposes for indefinite use, whereas commercial customers must sign a customer agreement for a six-month, limited-use license to evaluate the technology.
* SBI is fully licensed to distribute PiggyBac vectors as a partnership with Hera BioLabs, Inc.
References
How It Works
How It Works
The PiggyBac Transposon System’s Cut-and-Paste Mechanism
The efficient PiggyBac Transposon System uses a cut-and-paste mechanism to transfer DNA from the PiggyBac Vector into the genome. If only temporary genomic integration is desired, the Excision-only PiggyBac Transposase can be transiently expressed for footprint-free removal of the insert, resulting in reconstitution of the original genome sequence.
Figure 1. The PiggyBac Transposon System’s cut-and-paste mechanism.
- The Super PiggyBac Transposase binds to specific inverted terminal repeats (ITRs) in the PiggyBac Cloning and Expression Vector and excises the ITRs and intervening DNA.
- The Super PiggyBac Transposase inserts the ITR-Expression Cassette-ITR segment into the genome at TTAA sites.
- The Excision-only Super PiggyBac Transposase can be used to remove the ITR-Expression Cassette-ITR segment from the genome, for footprint-free removal
Tightly-controlled, inducible gene expression
Get robust, titratable gene expression with low background using SBI’s cumate-inducible vectors. These vectors take advantage of CymR, a repressor that binds to cumate operator sequences (CuO) with high affinity in the absence of cumate, a non-toxic small molecule. Providing much lower background expression than similar systems, SBI’s cumate-inducible vectors can provide up to 32-fold induction of gene expression.
- Robust—increase expression up to 32-fold
- Adjustable—tune expression levels by titrating the amount of cumate
- Reversible—turn expression on, then off, then on again
- Powerful—suitable for in vivo applications