RESEARCH HIGHLIGHT: Minicircle DNA breathes life into airway-delivered gene therapy development
Minicircle DNA from System Biosciences is fueling the development of gene therapy approaches for lung diseases.
One of the most exciting applications for minicircle DNA is in gene therapy, where getting safe and efficient transgene expression has been a major challenge to the field. While lentiviral vectors are widely used for mammalian gene expression in research applications, concerns about the safety of using viral vectors as human therapeutics has limited their use in gene therapy. Minicircle DNA is an attractive alternative as it is free of viral and bacterial sequences and can express transgenes for longer periods of time than plasmid DNA. Low toxicity and immunogenicity are especially important for applications that require repeated, long-term dosing, such as when treating short-lived cells like lung epithelial cells.
In a recent paper from University College London, Munye, et al., (2016)1, provide the first demonstration of how minicircle DNA enables enhanced and longer-lasting transgene expression in the lung than plasmid DNA, and paves the way for airway-delivered gene therapy approaches.
“In this study we have shown that LED-1 formulations containing minicircle DNA can provide greater and more persistent transgene expression which could increase the prospect of successful gene therapy for airway diseases.”1
The Challenges: Developing safe and effective gene therapy has been hampered by a number of challenges, including the ability to deliver transgenes to the cells that need them and getting sufficient levels of gene expression from a no/low immunogenicity and toxicity vector.
The Current State: Munye, et al, 1 have been pioneering a gene therapy approach for lung diseases such as cystic fibrosis, primary ciliary dyskinesia (PCD), surfactant protein B (SPB)-deficiency, and alpha 1-antitrypsin (A1AT)–deficiency. One advantage to focusing on genetic lung diseases is the relatively easy accessibility of the target cells, which can be reached using an inhaler or nebulizer. In addition, airway delivery may be useful as a general method for delivering gene therapies.
They’ve already developed a lipid/peptide formulation that is capable of delivering a plasmid payload to target cells in the lung when delivered via nebulizer to mouse and pig animal models.
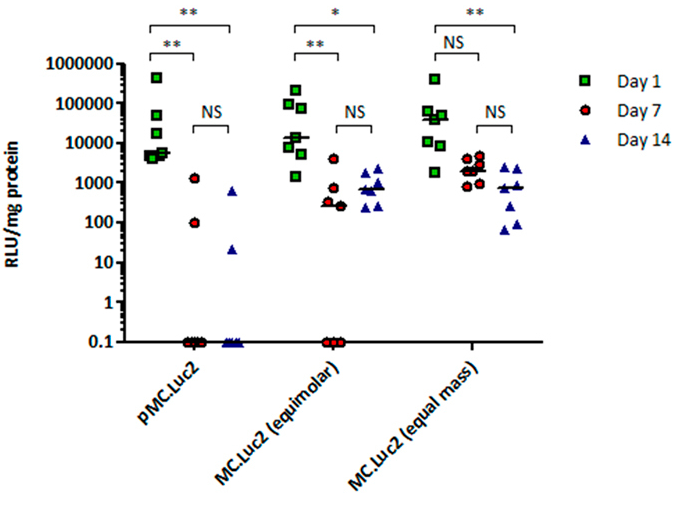
The Advances: In their paper, “Minicircle DNA Provides Enhanced and Prolonged Transgene Expression Following Airway Gene Transfer,” Munye, et al, 1 move on to optimizing expression of the transgene delivered by their lipid/peptide formulation. By moving from plasmid DNA to minicircle DNA, they were able to get longer-lasting expression of a reporter transgene in both cell and mouse models, paving the way for effective delivery and expression of a therapeutic transgene.
SBI Product Used*: Munye, et al, 1 cloned their reporter gene into the pMC.BESPX-MCS2 Empty Parental Minicircle Cloning Vector. Note that while academic researchers may clone and produce their own minicircle vectors, licensing agreements limit the use of this system by for-profit organizations. However, SBI is one of the few companies licensed to clone and produce custom minicircle plasmids—visit the Minicircle Services page to learn more. *Purchased from Cambridge Biosciences, one of SBI’s distributors in the UK and Ireland.
References
- Munye, et al. Minicircle DNA Provides Enhanced and Prolonged Transgene Expression Following Airway Gene Transfer. Sci Rep. March 2016; 6: 23125. PMCID: PMC4792149.