AAVanced™ Concentration Reagent
- Easy to use, single reagent
- Isolate rAAV directly from media
- No cell lysates or centrifugation
- Save time and cost-effective
- Compatible with all serotypes
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| AAV100A-1 | AAVanced Concentration Reagent | 100 mL | $479 |
|
||||
| AAV110A-1 | AAVanced Concentration Reagent | 250 mL | $962 |
|
||||
Overview
Overview
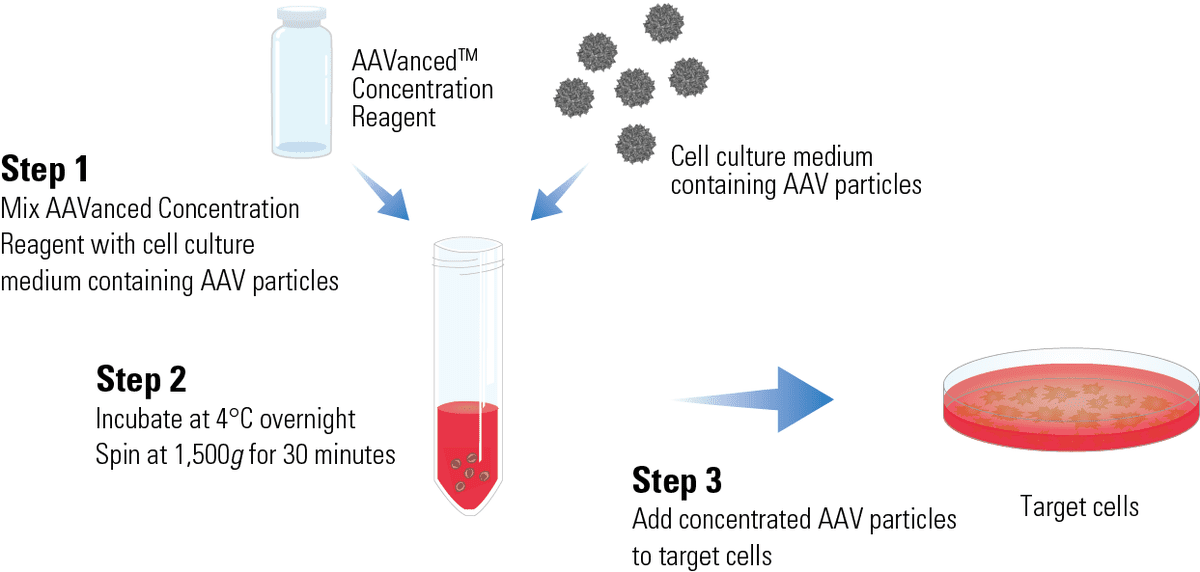
Streamline gene therapy development and other AAV-based applications
With a simple one-step protocol to concentrate AAV particles from media, you can streamline your gene therapy development and other projects that rely on AAV transduction.
- Easy to use, single reagent
- Isolate rAAV directly from media
- No cell lysates or centrifugation
- Save time and cost-effective
- Compatible with all serotypes
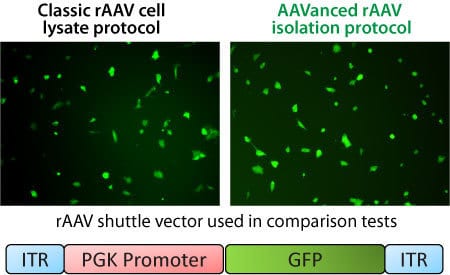
Figure 1. AAVanced Concentration Reagent delivers AAV particle preps with similar titers as AAV particles prepared using conventional methods. rAAV was packaged with a PGK-GFP expression shuttle vector. The rAAV particles were isolated from packaging cell media either by the conventional method—cell lysis freeze-thaw with density gradient ultracentrifugation—or with the AAVanced Concentration Reagent. Equal amounts of rAAV were then added to HEK293 cells to test for infectivity based on GFP expression. Representative images of GFP expressing cells 10-days after infection show similar levels of infectivity.
How It Works
Supporting Data
Supporting Data
Demonstrably high titer with rAAV isolated using AAVanced Concentration Reagent
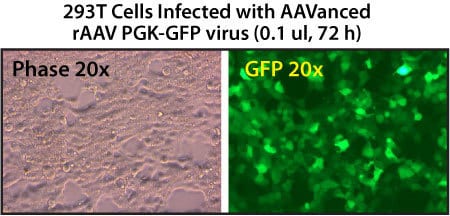
Figure 2. rAAV particles isolated using AAVanced Concentration Reagent are present at high copy numbers in 293T cells. A small volume of the rAAV virus isolated using AAVanced Concentration Reagent was used to infect 3×105 293T cells in a 24-well culture dish. The cells were imaged after 72-hours and rAAV copy numbers quantitated by qPCR. The copy numbers measured for the PGK-GFP rAAV virus were quite high at 2×1013 copies/mL.
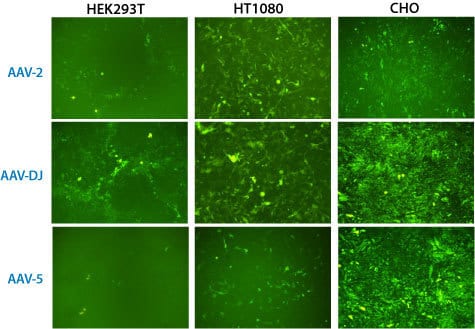
Figure 3. AAVanced Concentration Reagent works with multiple serotypes in vitro. The same rAAV shuttle vector with a PGK promoter driving GFP expression was packaged in parallel into three different AAV serotypes: AAV-2, AAV-DJ and AAV-5. After 48-hours, media was collected and rAAV particles isolated using the AAVanced Concentration Reagent. Each rAAV particle pellet was resuspended in 100 mL of sterile PBS, and then 1 mL of the rAAV suspensions were added to three different cell lines (HEK293T, HT1080 and CHO) to validate transduction efficiencies. The infected cells were imaged for GFP expression 6-days after addition of the isolated rAAV particles. All serotypes of rAAV were able to infect the three different cell types tested, demonstrating the broad compatibility of AAVanced Concentration Reagent.
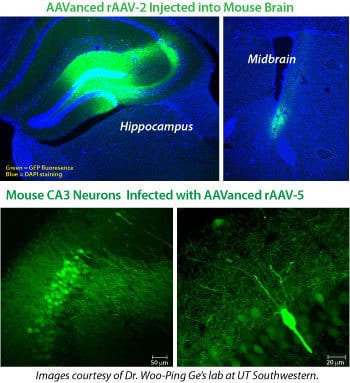
Figure 4. rAAV particles isolated using AAVanced Concentration Reagent work in vivo. (Upper panels) Representative images of hippocampal and midbrain sections from 6-week old C57 mice injected with AAV virus (ITR-PGK-GFP-ITR) concentrated using AAVanced Concentration Reagent. Approximately 1.5 µl of concentrated AAV virus (AAV-2 serotype) was delivered to hippocampal and midbrain regions by stereotaxic injection. Three weeks after virus injection, the animals were perfused with paraformaldehyde, fixed, and brains specimen were sectioned to 40 micron slices before imaging. The presence of robust GFP expression demonstrates successful delivery of the rAAV vector.
(Lower panels) Representative images of sections of mouse CA3 neurons in the hippocampus from 2-month old C57 mice injected with AAV-5 virus (ITR-PGK-GFP-ITR) concentrated using AAVanced Concentration Reagent. Approximately 1.5 µl of concentrated rAAV-5 virus was delivered to cortical regions by stereotaxic injection. Two-weeks after virus injection, animals were perfused by paraformaldehyde, fixed and brains were sectioned to 70 micron slices before imaging. The presence of robust GFP expression demonstrates successful delivery of the rAAV vector.
FAQs
Resources
Related Products
Citations
Products
| Catalog Number | Description | Size | Price | Quantity | Add to Cart | |||
|---|---|---|---|---|---|---|---|---|
| AAV100A-1 | AAVanced Concentration Reagent | 100 mL | $479 |
|
||||
| AAV110A-1 | AAVanced Concentration Reagent | 250 mL | $962 |
|
||||
Overview
Overview
Streamline gene therapy development and other AAV-based applications
With a simple one-step protocol to concentrate AAV particles from media, you can streamline your gene therapy development and other projects that rely on AAV transduction.
- Easy to use, single reagent
- Isolate rAAV directly from media
- No cell lysates or centrifugation
- Save time and cost-effective
- Compatible with all serotypes

Figure 1. AAVanced Concentration Reagent delivers AAV particle preps with similar titers as AAV particles prepared using conventional methods. rAAV was packaged with a PGK-GFP expression shuttle vector. The rAAV particles were isolated from packaging cell media either by the conventional method—cell lysis freeze-thaw with density gradient ultracentrifugation—or with the AAVanced Concentration Reagent. Equal amounts of rAAV were then added to HEK293 cells to test for infectivity based on GFP expression. Representative images of GFP expressing cells 10-days after infection show similar levels of infectivity.
Supporting Data
Supporting Data
Demonstrably high titer with rAAV isolated using AAVanced Concentration Reagent
Figure 2. rAAV particles isolated using AAVanced Concentration Reagent are present at high copy numbers in 293T cells. A small volume of the rAAV virus isolated using AAVanced Concentration Reagent was used to infect 3×105 293T cells in a 24-well culture dish. The cells were imaged after 72-hours and rAAV copy numbers quantitated by qPCR. The copy numbers measured for the PGK-GFP rAAV virus were quite high at 2×1013 copies/mL.
Figure 3. AAVanced Concentration Reagent works with multiple serotypes in vitro. The same rAAV shuttle vector with a PGK promoter driving GFP expression was packaged in parallel into three different AAV serotypes: AAV-2, AAV-DJ and AAV-5. After 48-hours, media was collected and rAAV particles isolated using the AAVanced Concentration Reagent. Each rAAV particle pellet was resuspended in 100 mL of sterile PBS, and then 1 mL of the rAAV suspensions were added to three different cell lines (HEK293T, HT1080 and CHO) to validate transduction efficiencies. The infected cells were imaged for GFP expression 6-days after addition of the isolated rAAV particles. All serotypes of rAAV were able to infect the three different cell types tested, demonstrating the broad compatibility of AAVanced Concentration Reagent.
Figure 4. rAAV particles isolated using AAVanced Concentration Reagent work in vivo. (Upper panels) Representative images of hippocampal and midbrain sections from 6-week old C57 mice injected with AAV virus (ITR-PGK-GFP-ITR) concentrated using AAVanced Concentration Reagent. Approximately 1.5 µl of concentrated AAV virus (AAV-2 serotype) was delivered to hippocampal and midbrain regions by stereotaxic injection. Three weeks after virus injection, the animals were perfused with paraformaldehyde, fixed, and brains specimen were sectioned to 40 micron slices before imaging. The presence of robust GFP expression demonstrates successful delivery of the rAAV vector.
(Lower panels) Representative images of sections of mouse CA3 neurons in the hippocampus from 2-month old C57 mice injected with AAV-5 virus (ITR-PGK-GFP-ITR) concentrated using AAVanced Concentration Reagent. Approximately 1.5 µl of concentrated rAAV-5 virus was delivered to cortical regions by stereotaxic injection. Two-weeks after virus injection, animals were perfused by paraformaldehyde, fixed and brains were sectioned to 70 micron slices before imaging. The presence of robust GFP expression demonstrates successful delivery of the rAAV vector.